(CDC/ Paul I. Howell, MPH; Prof. Frank Hadley Collins)
PHILADELPHIA – Dr. Keith Hamilton took his turn in the patient chair and braced for the sting of an experimental Zika vaccine.
The injection was the easy part. Next, a nurse jabbed three tiny needles in his upper arm with a device that delivered two electrical jolts strong enough to flex muscle. He said it felt like a needle piercing his arm, again and again.
Dr. Hamilton, an infectious-diseases doctor, was on a break from his rounds to volunteer in a landmark trial of a next-generation vaccine at the University of Pennsylvania’s medical school.
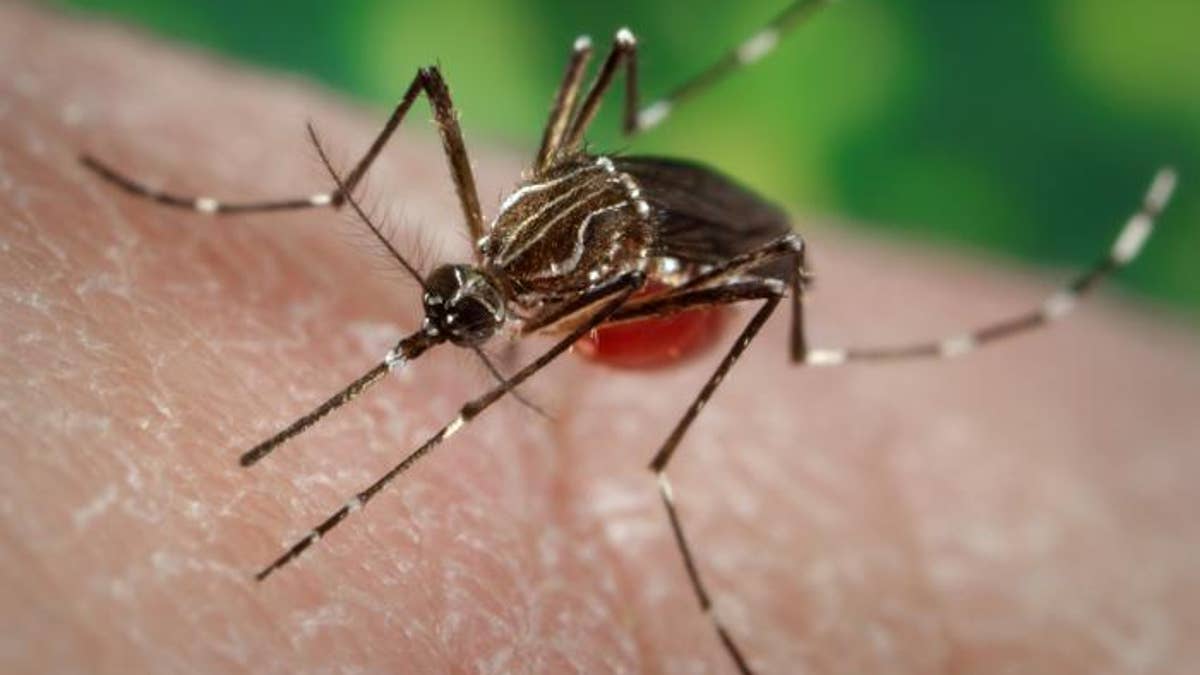
The Zika epidemic is accelerating work on this and other experimental DNA vaccines, which could turn out to be America’s best defense against infectious-disease outbreaks that now spread around the world with alarming speed, fueled by rising populations and global travel. These vaccines, made with synthetic DNA, can be developed and manufactured quickly.
Researchers in the U.S. and Canada have injected dozens of volunteers in the past few months with two competing DNA vaccines intended to provide immunity to the Zika virus. The mosquito-borne virus has caused hundreds of birth defects, including brain damage, and fetal deaths, mostly in Brazil.
Inovio Pharmaceuticals Inc., which makes the vaccine in the trial here, is in a race to market its vaccine and accompanying “electroporation device,” a tool the size of an electric toothbrush that uses a jolt of electricity to help usher the firm’s DNA vaccine into human cells.








































