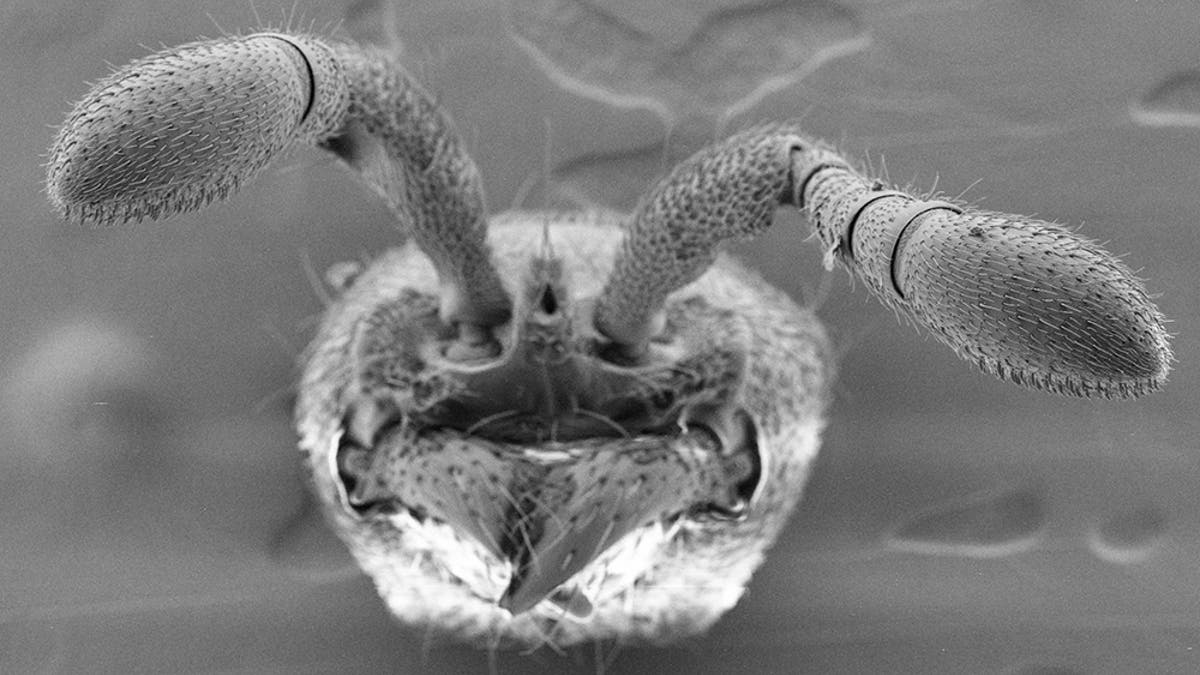
Ants use porous hairs on their antennae to detect pheromones. Scientists developed mutant ants lacking this ability. (Credit: Sean K. McKenzie/The Rockefeller University)
It may sound like a script for a science fiction movie, but scientists have created the world’s first mutant ants.
Two independent research teams have harnessed the gene editing technology CRISPR to genetically alter the ants. In one study, researchers at Rockefeller University modified a gene essential for sensing the pheromones that ants use to communicate. Experts say that the resulting deficiencies in the ants’ social behaviors and their ability to survive in a colony, sheds light on social evolution.
“It was well known that ant language is produced through pheromones, but now we understand a lot more about how pheromones are perceived,” said Daniel Kronauer, head of Rockefeller University’s Laboratory of Social Evolution and Behavior, in a statement. “The way ants interact is fundamentally different from how solitary organisms interact, and with these findings we know a bit more about the genetic evolution that enabled ants to create structured societies.”
GENE EDITING BREAKTHROUGH COULD PAVE WAY FOR PIG-TO-HUMAN ORGAN TRANSPLANTS
CRISPR, which has been compared to a pair of “molecular scissors,” lets scientists alter or replace specific sections of DNA.
Scientists used CRISPR to disrupt a gene known as Orco in the clonal raider ant, species Ooceraea biroi, but then faced the challenge of keeping the mutant ants alive. “We had to convince the colonies to accept the mutants. If the conditions weren’t right, the worker ants would stop caring for larvae and destroy them,” said Rockefeller University graduate fellow Waring Trible, in the statement. “Once the ants successfully made it to the adult phase, we noticed a shift in their behavior almost immediately.”
While ants typically travel single file, researchers noticed that the mutant ants couldn’t fall in line, along with other behavioral abnormalities.
DNA BREAKTHROUGH: SCIENTISTS REPAIR GENES IN HUMAN EMBRYOS TO PREVENT INHERITED DISEASES
The results of the study are published in the journal Cell.

This image shows a Harpegnathos saltator worker ant in the process of stinging a cricket to paralyze it and drag it into the nest as part of its hunting duties. (Credit: Brigitte Baella)
A separate study, also published in the journal Cell, saw scientists target the Orco gene in the Indian jumping ant, Harpegnathos saltator. Experts note that the Indian jumping ant is unlike other ant species because only the queen can mate and pass genes onto the next generation. However, any adult female worker of the species can become a “pseudo queen” in the queen’s absence.
The second study was led by researchers from New York University, NYU School of Medicine, Arizona State University, the University of Pennsylvania and Vanderbilt University.
Ant queens suppress the ability of female workers to mate and lay eggs, although if the queen is removed, the most aggressive females, after winning a series of antenna duels with rivals, can go on to lay eggs.
DNA DISCOVERY UNRAVELS THE MYSTERY OF EARLY GREEK CIVILIZATIONS
The study engineered three mutant ants to lack the Orco gene. Without the gene, females cannot process pheromones, making them less likely to engage in dueling.
"While ant behavior does not directly extend to humans, we believe that this work promises to advance our understanding of social communication, with the potential to shape the design of future research into disorders like schizophrenia, depression or autism that interfere with it," said Claude Desplan, professor at NYU's Department of Biology, and one of the report’s authors, in a statement.
In a third related study by the University of Pennsylvania, scientists injected the brain chemical corazonin into ants transitioning to become a pseudo-queen, which simulated worker-like hunting behaviors, while inhibiting ‘pseudo-queen’ behavior, such as dueling and laying eggs.
DNA DISCOVERY IDENTIFIES LIVING DESCENDANTS OF BIBLICAL CANAANITES
These results are also published in the journal Cell.
Gene editing has been generating plenty of buzz recently. Earlier this week, scientists announced the elimination of viruses in pigs that could be harmful to people, utilizing the CRISPR technology. The discovery could potentially lay the foundations for pig-to-human organ transplants.
GENE EDITING BREAKTHROUGH COULD PAVE WAY FOR PIG-TO-HUMAN ORGAN TRANSPLANTS
In another project, researchers used gene-editing to correct a disease-causing gene mutation in human embryos, preventing the mutation from passing to future generations. In the stunning discovery, a research team led by Oregon Health and Science University reported that embryos can fix themselves if scientists jump-start the process early enough.
Follow James Rogers on Twitter @jamesjrogers