The U.S. Food and Drug Administration approved a mechanical device that gives children extra time when their heart is failing.
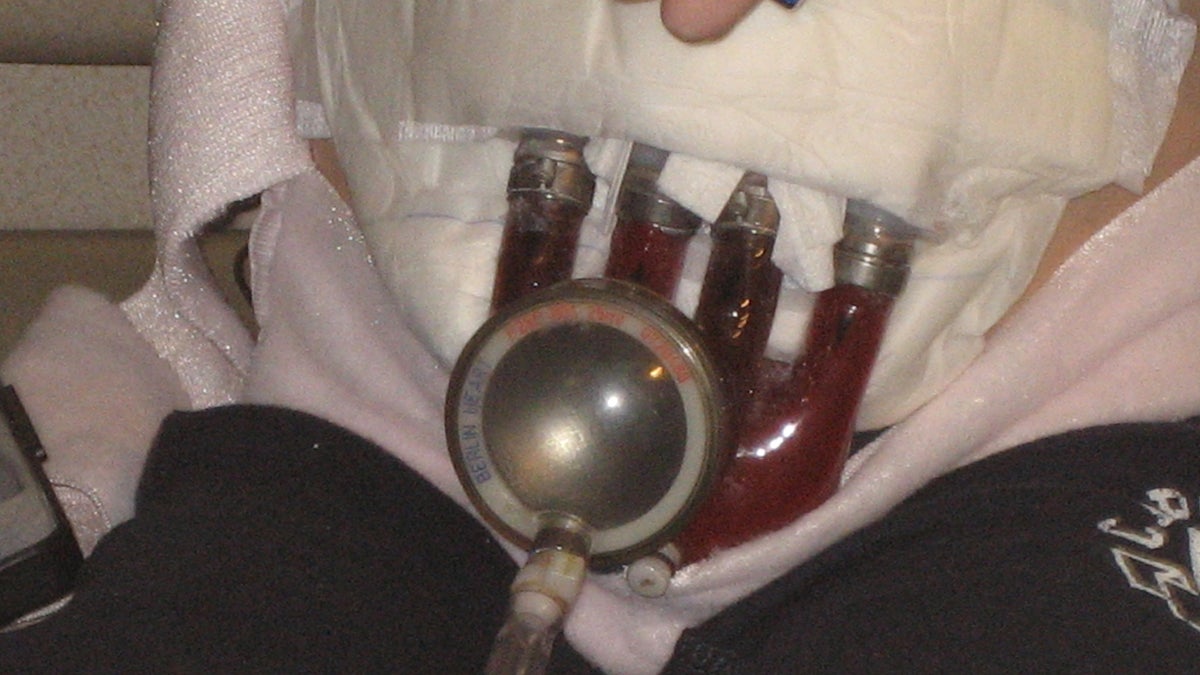
The external device, known as the Berlin Heart, works like a back-up pump to help pump blood throughout the body, said one doctor.
“Like any machine, the heart is a machine that can malfunction. When it’s malfunctioning, your body still depends on blood being pumped to every cell in your body,” said Dr. Robert Stewart of the Cleveland Clinic Children’s Hospital.
"So, this is really an auxiliary pump. This is a backup safety pump, and it essentially takes the blood as it is going into the pumping chamber, diverts it to here and pumps it to where it’s supposed to be going.”
The German company Berlin Heart AG was formed in 1997. Its three heart devices have been used widely across Europe for several years.
Prior to approval in the U.S., doctors had to apply for “compassionate” use of the device.
The device attaches to the heart’s ventricles by slim tubes and rests on the chest.
A small air hose at the device’s bottom hooks up to a machine, which provides the suction and pumping power needed to circulate blood throughout the body.
Children of all ages – newborns to teens – can use the device, which usually helps cardiomyopathy patients.
The device can work for several months while the child waits for heart transplant.
Stewart said the device keeps the child’s condition from weakening.
“So there are many, many cases where people have gone on to the Berlin heart on a ventilator, even on life support systems and get off all of those machines, gain weight and they’re just a totally different person at the time of transplant, and this makes all the difference in the world in terms of recovery,” Stewart said.
There is a risk of having a stroke while using the Berlin Heart, Stewart said, as clots can form, but blood thinners are used to decrease this risk.