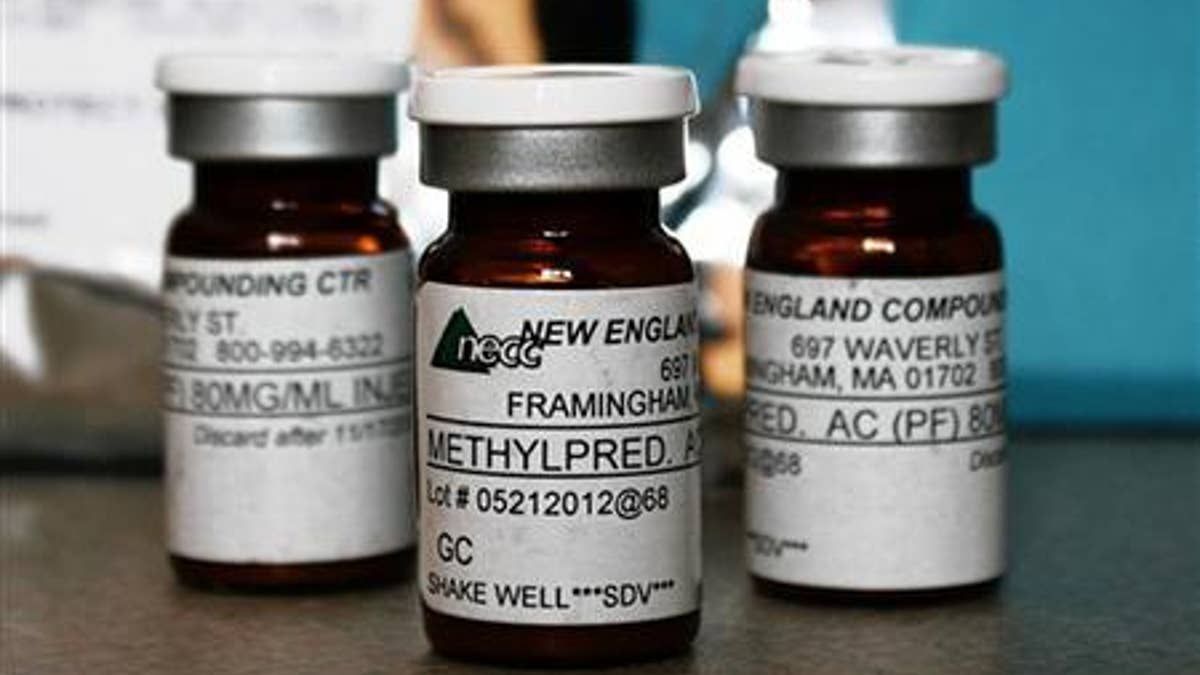
Vials of the steroid distributed by New England Compounding Center (NECC) - implicated in a meningitis outbreak - are pictured in this undated handout photo obtained by Reuters October 14, 2012. The outbreak has killed 15 people and put 14,000 at risk of contracting meningitis. REUTERS/Minnesota Department of Health/Handout.
The investigation into apparent contamination of injectable back pain medication from the New England Compounding Center has expanded to include two other products from the Massachusetts pharmacy.
While most of the reported infections involve fungal meningitis in patients who received epidural injections of methylprednisolone acetate, the Food and Drug Administration reports that one patient has developed possible meningitis symptoms after receiving a lower back injection of a different steroid from the NECC called triamcinolone acetone.
The FDA is also investigating reports that two heart transplant patients have developed fungal infections after receiving the NECC's cardioplegic solution, a medication used to paralyze the cardiac muscle to prevent injury during open heart surgery. Although the agency says it's too early to determine whether these apparent infections are linked to the medication, or some other cause.
Additionally, the FDA is concerned about the NECC's ophthalmic drugs used in injections or in connection with eye surgery, because they're processed under similar conditions. However, no infections associated with the NECC's ophthalmic drugs have been reported so far.
Public health officials are urging patients who have received doses of these NECC medications to contact a physician if they develop symptoms of fungal infection – including fever, swelling, discharge from the eye, chest pain or drainage from the surgical site.
Patients who received epidural steroid injections should also be alert for possible symptoms of fungal meningitis, which can include fever, headache, stiff neck, nausea, sensitivity to light or confusion.
As of Oct. 16, health officials had reported 214 fungal meningitis cases in 15 states, plus two cases involving non-meningitis fungal infections of peripheral joints (such as knees, hips, shoulders and elbows).
Sixteen deaths have been reported in connection with the fungal meningitis outbreak.