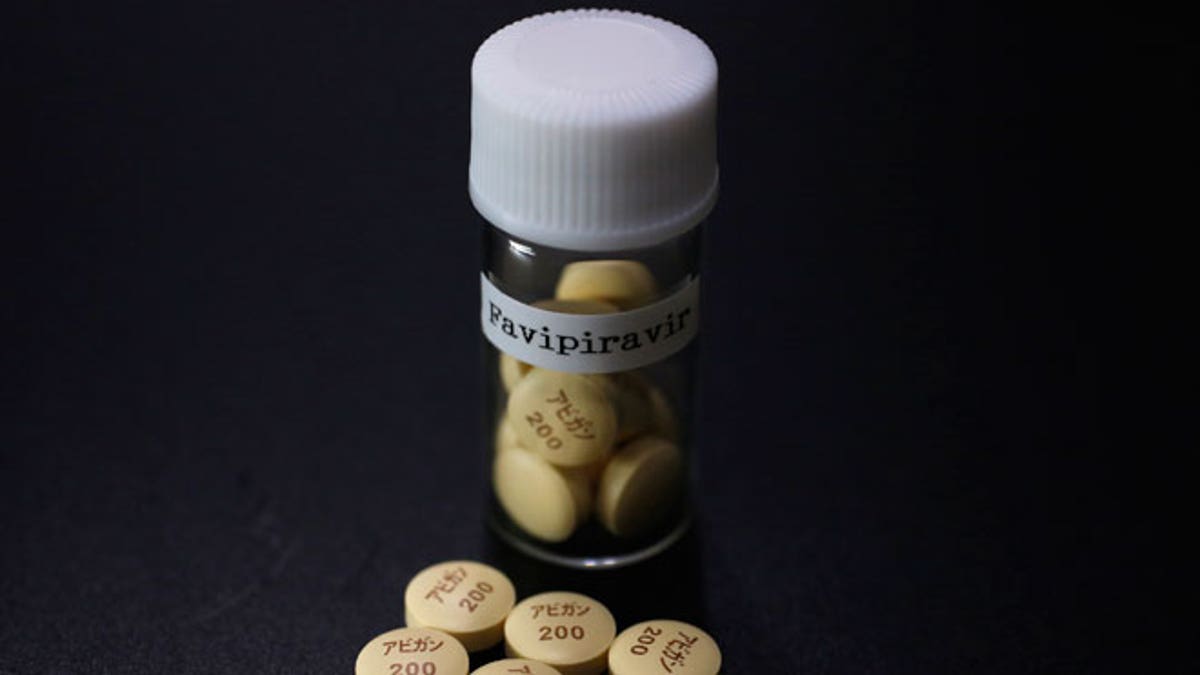
Tablets of Avigan (generic name : Favipiravir), a drug approved as an anti-influenza drug in Japan and developed by drug maker Toyama Chemical Co, a subsidiary of Fujifilm Holdings Co. are displayed during a photo opportunity at Fujifilm's headquarters in Tokyo October 22, 2014. Japan's Fujifilm Holdings Corp said on Monday it was expanding the production of its Avigan anti-influenza drug to reach an additional number of Ebola patients. France and Guinea plan to conduct clinical trials of Avigan 200 mg tablets, made by Fujifilm group company Toyama Chemical Co, in Guinea to treat Ebola in mid-November, Fujifilm said in a statement. (REUTERS/Issei Kato)
France said on Wednesday the first trials of a drug to treat Ebola in Guinea had been encouraging and appeared to accelerate the recovery process of patients, President Francois Hollande's office said in a statement.
Trials of an experimental Japanese drug - Avigan, or favipiravir - developed by Toyama Chemical, a subsidiary of Japan's Fujifilm, began at a treatment center in Guinea in mid-December.
"The results are encouraging. They show a fall in the number of deaths in adults and teenagers. The recovery process is accelerated," Hollande's office said after he met doctors working on tackling Ebola. It added the treatment was given to 80 patients, adults and children.







































