Move Back
ADVERTISEMENT
Skip- Published11 Images
H1N1: One Year Later
This week marks one year since the H1N1 flu virus was first identified in April of 2009.
![H1N1: A Year Later]() In the months that followed the announcement of this new flu virus, health organizations across the world began to mobilize in an all out effort to contain H1N1, even declaring the first global flu pandemic in more than 40 years. But, looking back – we ask the question: Was this the pandemic that never was? Let’s start from the beginning.read moreiStockShare
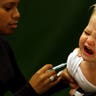
In the months that followed the announcement of this new flu virus, health organizations across the world began to mobilize in an all out effort to contain H1N1, even declaring the first global flu pandemic in more than 40 years. But, looking back – we ask the question: Was this the pandemic that never was? Let’s start from the beginning.read moreiStockShare![March 2009]() In the weeks before Americans were officially introduced to the H1N1 flu - signs of the virus began to raise red flags in Mexico and southern California. That’s where a total of three young children came down with upper respiratory infections. Soon after, health officials collected specimen samples. All three children recover, but this marks the beginning of a global health scare. Edgar Hernandez, 5, known as 'Patient Zero,' may be the first person to have contracted the novel H1N1 flu.read moreAFPShare
In the weeks before Americans were officially introduced to the H1N1 flu - signs of the virus began to raise red flags in Mexico and southern California. That’s where a total of three young children came down with upper respiratory infections. Soon after, health officials collected specimen samples. All three children recover, but this marks the beginning of a global health scare. Edgar Hernandez, 5, known as 'Patient Zero,' may be the first person to have contracted the novel H1N1 flu.read moreAFPShare![April 2009]() This is the month where things really started to take off. In the early part of April 2009, the Centers for Disease Control and Prevention examined those samples collected in March. It is at this time, they realize they’re looking at an “unsubtypable” influenza A virus - not reported previously anywhere in the world. At the same time, a death from this new “swine” flu strain is reported in Oaxaca, Mexico. Soon after, the CDC suspects human-to-human transmission when they identify a child from a neighboring county with the same novel H1N1 virus. Neither child has had any contact with pigs. Several days later, the virus turns up in two children in Texas. From there, it becomes clear to the agency that a multinational outbreak is in progress. By the end of April, U.S. cases rise to 20 prompting the government to declare a public health emergency.read moreReutersShare
This is the month where things really started to take off. In the early part of April 2009, the Centers for Disease Control and Prevention examined those samples collected in March. It is at this time, they realize they’re looking at an “unsubtypable” influenza A virus - not reported previously anywhere in the world. At the same time, a death from this new “swine” flu strain is reported in Oaxaca, Mexico. Soon after, the CDC suspects human-to-human transmission when they identify a child from a neighboring county with the same novel H1N1 virus. Neither child has had any contact with pigs. Several days later, the virus turns up in two children in Texas. From there, it becomes clear to the agency that a multinational outbreak is in progress. By the end of April, U.S. cases rise to 20 prompting the government to declare a public health emergency.read moreReutersShare![May 2009]()
![June 2009]() For the first time in more than 40 years, The World Health Organization declares a global flu pandemic . "Further spread of the disease is inevitable," WHO Director-General Dr. Margaret Chan said at a media briefing on June 11, 2009. At the time of the announcement, more than 70 nations had reported nearly 30,000 cases of the virus, including 18,000 in the U.S. The death toll reaches 144. In response to the pandemic announcement, the Department of Health and Human Services secretary, Kathleen Sebelius states: “What this declaration does do is remind the world that flu viruses like H1N1 need to be taken seriously…we need to start preparing now in order to be ready for a possible H1N1 immunization campaign starting in late September.”read moreAFPShare
For the first time in more than 40 years, The World Health Organization declares a global flu pandemic . "Further spread of the disease is inevitable," WHO Director-General Dr. Margaret Chan said at a media briefing on June 11, 2009. At the time of the announcement, more than 70 nations had reported nearly 30,000 cases of the virus, including 18,000 in the U.S. The death toll reaches 144. In response to the pandemic announcement, the Department of Health and Human Services secretary, Kathleen Sebelius states: “What this declaration does do is remind the world that flu viruses like H1N1 need to be taken seriously…we need to start preparing now in order to be ready for a possible H1N1 immunization campaign starting in late September.”read moreAFPShare![July-August 2009]() In July and August, progress is really starts to pick up on the development and testing of an H1N1 vaccine. The first human trial is held in Australia, and shortly after clinical trials begin in the U.S. that involve high-risk groups including children, pregnant women, people with asthma, and people infected with HIV. As students begin to return to school and college campuses across the country, a massive campaign is launched in an effort to educate people about how to prevent the spread of the virus. Still, cases among students begin to rise.read moreReutersShare
In July and August, progress is really starts to pick up on the development and testing of an H1N1 vaccine. The first human trial is held in Australia, and shortly after clinical trials begin in the U.S. that involve high-risk groups including children, pregnant women, people with asthma, and people infected with HIV. As students begin to return to school and college campuses across the country, a massive campaign is launched in an effort to educate people about how to prevent the spread of the virus. Still, cases among students begin to rise.read moreReutersShare![September 2009]() In September, the FDA announces the approval of four vaccines that would be distributed nationwide. Trial results of the vaccine proved one dose to be effective in protecting healthy adults and older children, and two doses for children under 9-years-old. Research later showed that only one dose of vaccine was needed to provide immunity, whereas most planning had been built around two doses being required. Soon after, questions surfaced about the vaccine’s link to the pharmaceutical industry after companies like GlaxoSmithKline and Sanofi-Aventis profited from producing H1N1 vaccine. Other doubts about the distribution of the vaccine criticized the United Nations for failing to ensure that developing countries received a fair share of vaccines developed against the virus.read moreReutersShare
In September, the FDA announces the approval of four vaccines that would be distributed nationwide. Trial results of the vaccine proved one dose to be effective in protecting healthy adults and older children, and two doses for children under 9-years-old. Research later showed that only one dose of vaccine was needed to provide immunity, whereas most planning had been built around two doses being required. Soon after, questions surfaced about the vaccine’s link to the pharmaceutical industry after companies like GlaxoSmithKline and Sanofi-Aventis profited from producing H1N1 vaccine. Other doubts about the distribution of the vaccine criticized the United Nations for failing to ensure that developing countries received a fair share of vaccines developed against the virus.read moreReutersShare![October 2009]() At the start of October, 26 states had reported H1N1 flu activity, which jumped to 46 by the end of the month. Ninety-five deaths have been reported in children and teens from H1N1. President Obama proactively declares the H1N1 flu a “National Emergency,” which some blame for widespread panic over the virus and a surge of people rushing to get vaccinated, with limited doses of the vaccine available. The CDC claimed by the end of October, 14.1 million doses were available for ordering by the states.read moreReutersShare
At the start of October, 26 states had reported H1N1 flu activity, which jumped to 46 by the end of the month. Ninety-five deaths have been reported in children and teens from H1N1. President Obama proactively declares the H1N1 flu a “National Emergency,” which some blame for widespread panic over the virus and a surge of people rushing to get vaccinated, with limited doses of the vaccine available. The CDC claimed by the end of October, 14.1 million doses were available for ordering by the states.read moreReutersShare![November 2009]() By the beginning of November, the H1N1 virus has been reported in 48 states and the ratio of affect on young people to adults is growing more disproportionate. Because of a reported shortage of vaccines, the government and CDC make a decision to ration the vaccine to selected high-risk groups, including pregnant women, children 6 months to 18 years old, young adults 19 to 24 years old, and those with a higher risk of medical complications from influenza. The number of deaths from H1N1 has risen to 198, but the CDC continues to cite increasingly high death rates - topping out at 500 - of unreported cases of H1N1, which were never confirmed. The FDA approves a fifth and final H1N1 vaccine, and approves an existing vaccine for adults to be used on infants and children.read more
By the beginning of November, the H1N1 virus has been reported in 48 states and the ratio of affect on young people to adults is growing more disproportionate. Because of a reported shortage of vaccines, the government and CDC make a decision to ration the vaccine to selected high-risk groups, including pregnant women, children 6 months to 18 years old, young adults 19 to 24 years old, and those with a higher risk of medical complications from influenza. The number of deaths from H1N1 has risen to 198, but the CDC continues to cite increasingly high death rates - topping out at 500 - of unreported cases of H1N1, which were never confirmed. The FDA approves a fifth and final H1N1 vaccine, and approves an existing vaccine for adults to be used on infants and children.read more![December 2009]() By December, H1N1 flu activity settles down with just 14 states reporting cases. Doctors visits and hospitalizations due to flu-like symptoms decrease. Even though H1N1 infection has decreased, the number of vaccinations produced continues to increase, allowing the general public to start getting vaccinated. The Department of Health and Human Services (HHS) continued to encourage the public to get vaccinated and warns of a possible winter outbreak. By the end of the year, it is estimated that 71 million H1N1 flu vaccines went unused. More anti-flu campaigns launch, including webcasts with the former president of the American Medical Association, and PSAs from the HHS.read moreReutersShare
By December, H1N1 flu activity settles down with just 14 states reporting cases. Doctors visits and hospitalizations due to flu-like symptoms decrease. Even though H1N1 infection has decreased, the number of vaccinations produced continues to increase, allowing the general public to start getting vaccinated. The Department of Health and Human Services (HHS) continued to encourage the public to get vaccinated and warns of a possible winter outbreak. By the end of the year, it is estimated that 71 million H1N1 flu vaccines went unused. More anti-flu campaigns launch, including webcasts with the former president of the American Medical Association, and PSAs from the HHS.read moreReutersShare![Looking Ahead]() Over the past year, the CDC , WHO, and U.S. government have claimed both successes and failures in stopping the spread of H1N1. Although an effective vaccine was developed that 80 million people utilized, the shortage of domestically produced vaccines caused many patients to go without. The World Health Organization conceded shortcomings in its handling of the H1N1 flu pandemic, including a failure to communicate uncertainties about the new virus as it swept around the globe. The CDC is currently in preparation to improve response to next year's flu season by increasing production of vaccines domestically, expanding school vaccination programs, and increasing vaccine uptake in communities with a history of low vaccination rates.read moreiStockShare
Over the past year, the CDC , WHO, and U.S. government have claimed both successes and failures in stopping the spread of H1N1. Although an effective vaccine was developed that 80 million people utilized, the shortage of domestically produced vaccines caused many patients to go without. The World Health Organization conceded shortcomings in its handling of the H1N1 flu pandemic, including a failure to communicate uncertainties about the new virus as it swept around the globe. The CDC is currently in preparation to improve response to next year's flu season by increasing production of vaccines domestically, expanding school vaccination programs, and increasing vaccine uptake in communities with a history of low vaccination rates.read moreiStockShare- Published11 Images
H1N1: One Year Later
This week marks one year since the H1N1 flu virus was first identified in April of 2009.
Move Forward
- H1N1: One Year Later





Thumbnail View
Image 0 of 11